Despite reduced levels of private financing, a near absence of major IPOs and persistent layoffs over the past two years, the biopharma industry, and biomanufacturing in particular, continue to be an attractive industry target for economic developers.
Despite some well-publicized downsizings in the aftermath of the pandemic, biopharma employment growth has tended to trend higher than overall private sector job generation for a number of years running. According to a recent report from TEConomy and the Biotechnology Innovation Organization, U.S. bioscience job creation increased by 11 percent between 2018 and 2021, while the private sector overall experienced a loss of nearly two percent during the same period. These jobs are often highly skilled and well paid, and because the industry is labor-intensive, it remains a very attractive target.
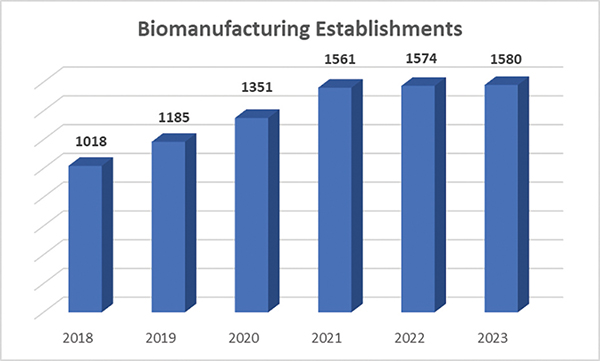
Within the larger pharmaceutical sector, the number of U.S. biomanufacturing plants, where so many of our newest medicines and other therapies are produced for clinical trials and commercial sale, has grown by more than 50 percent in recent years, according to Pharmaceutical Research and Manufacturers (PhRMA), an industry association.
All of this activity generates a very strong multiplier effect, estimated by PhRMA at $1.4 trillion of economic impact (or 3.7 percent of total U.S. gross domestic product), and more than 4.4 million highly skilled jobs across the country.
Ariel Gruswitz, Vice President of Facility Logix, a life science consulting partner of Biggins Lacy Shapiro & Company (BLS & Co.), observed, “This growth, which has traditionally been concentrated in the mature biopharma clusters on both coasts, is emerging in newer geographies.”
While California, Massachusetts and North Carolina still lead the production of large molecule medicines and other advanced therapies, Facility Logix and BLS & Co. have seen increased biomanufacturing interest and investment in states such as Maryland, Delaware, Illinois, Texas, Ohio and Colorado. Other legacy clusters such as Pennsylvania and New Jersey continue their traditional dominance in small molecule manufacturing as they pivot to more advanced therapies as well.
Near-Term Challenges
Of course, the industry is not without its challenges, most notably, the newly announced Medicare price negotiations for the first 10 prescription drugs, national drug shortages, manufacturing quality control issues, surges in demand for new obesity medicines and even natural disasters. For example, in July 2023, a tornado struck Pfizer’s manufacturing plant in Rocky Mount, North Carolina.
The resulting damage to the facility, which supplies eight percent of all sterile injectable medicines used in U.S. hospitals, forced Pfizer to ration the supply of 12 injection products, which were to be distributed only through emergency orders “due to their high medical need.” As this article was being written, the website Fierce Pharma reported that Pfizer has “signaled its intention to restart manufacturing at Rocky Mount by the fourth quarter of 2023.”
The other major dynamic impacting the biopharma industry has been the slowdown in the capital markets over the past several years. A recent report by HSBC noted a 40 percent drop-off in early-stage funding between 2022 and 2023 as investors look for safer bets in known entities. And, according to Jones Lang LaSalle’s 2022 Life Sciences Outlook, VC funding for bioscience companies reached historic highs of nearly $80 billion in recent years but has begun to level out.
Such funding constraints have had a predictable effect on hiring. The Fierce Biotech Layoff Tracker has reported a total of 119 layoffs through the middle of August 2023 — the same as 2022’s entire count.

However, the Fierce Biotech data reveal that these cutbacks have largely affected smaller pre-clinical and clinical trial drug companies hoping to preserve cash, withstand failures or survive collapsed mergers and partnerships.
While big bio was not immune, relatively few of the recent cutbacks at large pharmaceutical companies appear to have affected their manufacturing operations. Among them, perhaps the most notable were Genentech’s intentions to close a 265-person production unit at its South San Francisco, California location (the company has long had plans to migrate commercial manufacturing to other sites in their global network) and Cepheid’s planned 1,500 job cuts at its facilities that had been making COVID-19 test kits.
Industry Resilience
At the same time however, the promise of new medicines from a few big pharma companies has driven the industry to even greater heights. Eli Lilly, which recently became the largest pharmaceutical company in the world by market capitalization, announced over $6 billion in new manufacturing investments in Indiana and North Carolina since 2020. Novo Nordisk’s market capitalization recently surpassed the total GDP of its home country of Denmark.
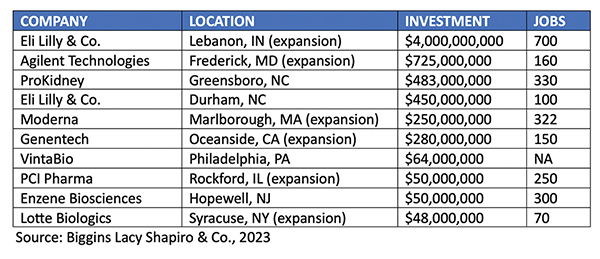
The first half of 2023 has witnessed continued demand for new production sites for biologics medicines. BLS & Company’s tracking data base has identified these major U.S. biomanufacturing announcements in the first half of 2023 alone.
Recent Biomanufacturing Announcements (Q1 and Q2 2023)
 Also, the Biden administration has made it known that reshoring biomanufacturing is a priority. The EDA Tech Hubs Phase II funding and the biotechnology and biomanufacturing initiative announced by the White House last fall are expected to add further impetus to the industry’s growth.
Also, the Biden administration has made it known that reshoring biomanufacturing is a priority. The EDA Tech Hubs Phase II funding and the biotechnology and biomanufacturing initiative announced by the White House last fall are expected to add further impetus to the industry’s growth.
According to Facility Logix’s Gruswitz, “Despite recent uncertainty of private capital availability, the federal government is signaling its strong support for significantly increasing domestic biomanufacturing capacity over the next 10 years as part of U.S. national and economic security goals.”
Thus, shaking off some recent challenges, the U.S. biopharma sector is expected to remain in expansion mode. BLS & Co. would advise life science companies contemplating new biomanufacturing locations or growth at existing sites, and economic developers eager to attract these high-quality jobs and investments, to become well-acquainted with the site selection criteria that typically drive these location decisions.
Site Selection Criteria
The critical success factors driving biomanufacturing location decisions generally fall into these six buckets:
• Access to specialized talent.
• An inventory of suitable properties.
• The presence of an existing (or developing) biomanufacturing cluster.
• A competitive cost structure.
• Available and usable incentives.
• The ability to know, and manage, risk.
Talent
Drug manufacturing remains a very labor-intensive operation requiring highly specialized skillsets. Drug product fill/finish facilities need process technicians trained to operate sensitive equipment in a cGMP clean room environment. An even more narrow set of skills is required for drug substance facilities producing active pharmaceutical ingredients, complex large molecule medicines and cutting-edge gene and cell therapies.
Key talent considerations should include:
• The availability of experienced bioprocessing, technical, analytical, engineering and managerial labor.
• The extent of the pipeline for new bioprocessing talent (focusing on undergraduate and graduate programs, two-year degree and certificate programs, etc.).
• The infrastructure to support continuing education, including customized training and open enrollment courses.
Real Estate
Facing long lead times for drug approvals, site selection, design and construction as well as plant commissioning and validation, medicine makers will be eager to launch commercial production as quickly as possible. Choosing the right location and site is one of the most predictable ways to shave even a few months from the overall timeline and can thus gain market share and achieve revenues that might otherwise be lost to competition.
Real estate criteria include:
• The size and configuration of the property and its ability to accommodate expansion.
• Proven ownership and control of the site or building, and the lack of any legal impediments to its sale.
• The listing price and the degree of competition (leverage) in the local property market.
• The extent to which additional investigations are necessary to vet any potential effects from environmental issues, wetlands, floodplains, subsurface geotechnical conditions, archeological and historical remains, etc.
• The availability of utilities to the site and the sufficiency of capacity in the local utility systems.
• The necessary land use approvals and entitlements (or a realistic timeline to achieve the same).
• The compatibility of adjacent land uses.
Cluster Benefits
The presence of other companies in the pharmaceutical, biotech and similar regulated industries can confer operational benefits such as access to talent and markets and the various types of support needed to operate an advanced industrial facility.
These variables include:
• Proximity to potential collaborators and partners.
• Proximity to suppliers of GMP products and services, including validation engineers, clean room suppliers, etc.
• The presence and density of establishments in the pharmaceutical and medicine manufacturing sectors.
• The presence of establishments in other highly regulated manufacturing industries such as veterinary medicines, cosmetics and nutraceuticals.
• The presence of active industry trade associations and similar organizations.
Costs
Costs are always a key site consideration for a biopharma manufacturing operation.
Factors to evaluate include:
• The one-time investment to stand up a new facility, including property acquisition and preparation, engineering and
design, construction, commissioning and validation.
• The ongoing costs of operation including labor, facility
operations (including utilities and maintenance), state and
local taxes, etc.
Incentives
Economic development incentives are a pricing tool employed by states and local jurisdictions to help offset some of the above costs and risks, and thus level the playing field. Incentives will vary by location, but typically include tax abatements, tax credits, cash grants, training assistance and infrastructure upgrades. Occasionally a prospect company will be offered free or discounted land or buildings.
Maximizing the financial benefit of these public funding tools must be balanced against the commitments that will be required in any incentives agreement. Important variables that should be considered include:
• The information and declarations that must be supplied in any incentives application.
• The approvals process and associated requirements for public disclosure.
• Any post-approval compliance/audit requirements.
• Any penalties or remedies imposed in the event of any failure to fulfill contractual requirements.
Risk
Risks can be manmade and/or naturally occurring. Some of the most significant threats that should be vetted as part of the site selection process include:
• Suppliers and vendor resilience.
• Logistics and transportation bottlenecks.
• Threats to vital infrastructure.
• Climatological risk, including increasing threats from rising water, storms, fire, etc.
• Threats to employee quality of life from crime, rising costs, etc.
• The state and local political climate and how policies might impact a company’s public standing and its ability to attract and retain a qualified workforce.
Other Operational Factors
The above list of key criteria is necessarily incomplete, as these factors must be tailored to the specific and individual requirements of each company and its site selection project. Other dynamics that could influence site selection might include:
• Community engagement and local acceptance of industrial development and growth
• Renewable energy mandates.
• State and local fiscal stability.
• Historical and trending labor relations.
Conclusion
The making of biopharmaceuticals is a highly advanced and complex form of manufacturing, requiring specialized skills, dedicated facilities, equipment and processes, and considerable investments of financial, intellectual and professional capital. The industry, which has lately been buffeted by the social, political and economic crosswinds of the post-pandemic era, still retains much of its attractiveness to those eager to grow and diversify their local economies and upskill their resident workforce and talent pipeline. Similarly, the makers of increasingly effective and valuable new medicines and therapies are highly motivated to locate their plants within states and regions that have proven their competitive capability to sustain operations over the long-term. When well executed, site selection enables a win-win that ensures that both the supply and demand sides of this equation are able to achieve their goals. T&ID


