 Batch processing has been the traditional way to manufacture pharmaceutical drugs for more than 50 years. Even with advances in technology, the underlying process to manufacture drugs has largely been the same, despite estimates that the pharmaceutical industry wastes up to $50 billion per year on it. Now a new, more efficient process called ‘continuous processing’ is beginning to catch on. This article explores the reasons for the transition and the impact it will have on drug development facilities across the U.S.
Batch processing has been the traditional way to manufacture pharmaceutical drugs for more than 50 years. Even with advances in technology, the underlying process to manufacture drugs has largely been the same, despite estimates that the pharmaceutical industry wastes up to $50 billion per year on it. Now a new, more efficient process called ‘continuous processing’ is beginning to catch on. This article explores the reasons for the transition and the impact it will have on drug development facilities across the U.S.
Batch Processing: Inefficient and Risky
Batch processing consists of a step-by-step process. Each “step” is completed in one room, for sanitary purposes, where a bulk material enters the space, is processed and then exits the room. Typically, testing of the partially completed product occurs in between each batch step, before the entire lot is tested again and released for sales and distribution.
Although the process works, it is extremely inefficient: Raw materials are produced at separate facilities around the world and shipped to processing facilities, which means significant downtime between steps.
Batch processing is also a risky way to manufacture drugs. The more time partially completed products spend between steps, the higher the risk for human error or contamination. As a result, pharmaceutical producers have sought out improved manufacturing processes to increase consumer safety and limit risks of recalls.
 A New Processing Method
A New Processing Method
Continuous processing – the same method that the automotive, food and electronics industries use to manufacture products – is growing in popularity as a method to manufacture pharmaceutical drugs in large volumes, particularly for tablet and pill products. It allows quality assurance officials to monitor large quantities of products at once — as opposed to one batch at a time. The continuous process sends raw materials through an uninterrupted, nonstop process until the final product is completed.
“Continuous processing, as the name might imply, is connection of all the process steps in sequence without stops, with in-line testing in real-time, which eliminates the need for batch testing of products,” said Michael Rooney, director of process engineering at Genesis, a Pennsylvania-based engineering firm that specializes in life science operations.
According to Rooney, for tablet and pill manufacturing, the continuous production process is designed to be vertically integrated in a multi-story operation, where the ingredients enter from the top and come out at the bottom as completed tablets or pills. Gravity moves the product through each processing step, eliminating the need for human contact during the process. This saves the company not only on labor costs, but also minimizes the risk of contamination of the product during the process.

In contrast, in a batch processing operation completed products must be stored for two weeks for final testing before products can be released to the market.
One method the pharmaceutical industry may choose to base a continuous manufacturing platform on what is called ‘Hot Melt Extrusion’ (HME). Primarily used in the plastic and food industries, HME is drawing attention from pharmaceutical quality heads for its ability to manufacture a wide variety of dosage formulations. This will aid in manufacturing drugs beyond tablets and pills and help handle poorly soluble APIs.
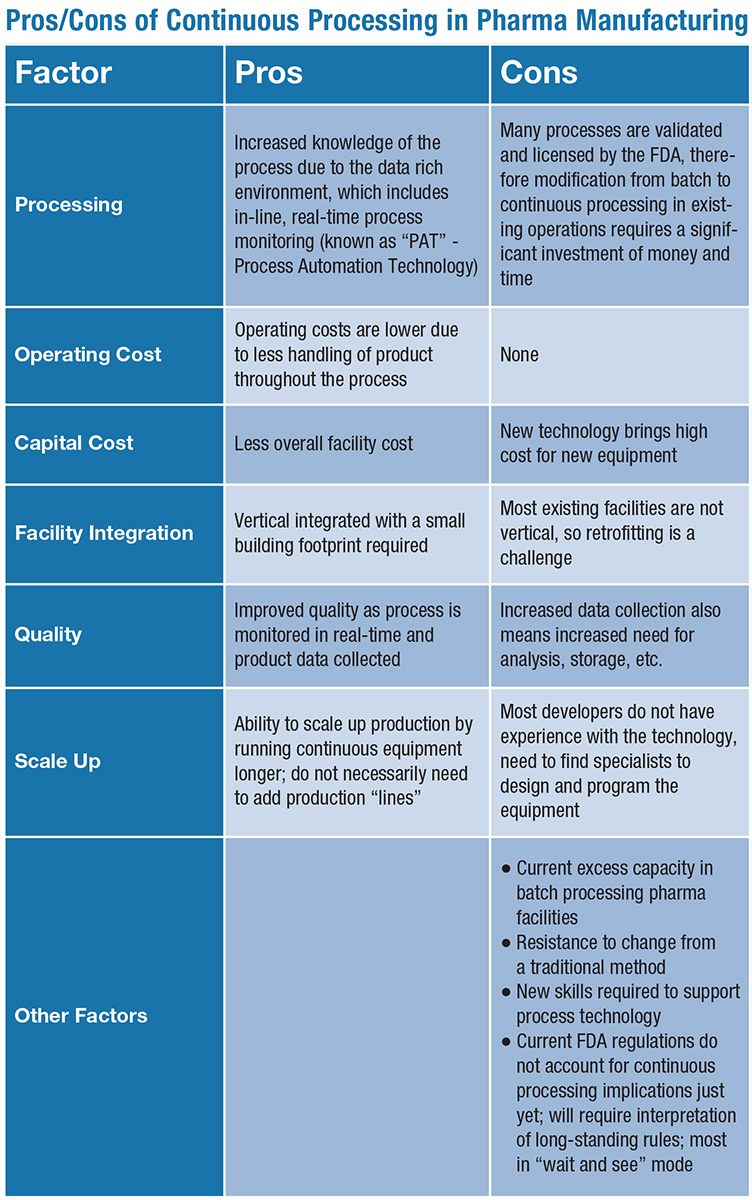
Batch vs. Continuous Processing
Although the numerous benefits of continuous processing are apparent, don’t expect all pharmaceutical manufacturers to abandon their batch processes just yet. While continuous processing is a more efficient way to manufacture drugs, many U.S. pharmaceutical manufacturing plants currently have excess capacity, meaning there is less urgency to transition from one method to another.
According to Rooney, the cost savings may not be great enough to justify the cost to transfer production processes from batch to continuous: the upfront costs for equipment are high, and the time required to re-register products with the FDA can be a deterrent. “However, most companies that want to reduce cost and increase overall product quality will try to apply continuous processing in the near term,” Rooney said.
In 2016, Johnson & Johnson’s Janssen drug unit converted from a batch to continuous process for production of HIV drug Prezista, after working on design of the new process for five years before obtaining FDA approval. The line at its Gurabo, Puerto Rico, plant became the first the FDA approved to switch to the new system. The new process requires less production space, while also shortening the production timeline from two weeks to one day. The real-time testing also improves the quality control and allows for immediate corrections, resulting in less waste if a product issue arises during the process.
As part of the Janssen announcement, Lawrence Yu, deputy director of the FDA’s Office of Pharmaceutical Quality was quoted as saying, “The FDA encourages others in the pharmaceutical industry to consider similar efforts.” Yu referenced the benefits of continuous processing being “faster” and “cheaper,” but even more important, reliable and safe.
The FDA is seeking to support production processes, including continuous manufacturing, that protect against regulatory problems, which should mean fewer drug shortages in the marketplace.
Despite these statements, the FDA has yet to update its official regulations to account for continuous processing. This has some existing manufacturers hesitant to convert existing facilities to continuous processing for fear of regulatory hurdles that cost time and money. However, other companies, such as Vertex in Boston, are constructing new plants designed to use continuous processing from the start.
Encouraging further growth of a standardized continuous processing method, Continuus Pharmaceuticals announced in November 2016 that it had been awarded a $4.4 million contract from the FDA. The contract tasks Continuus with developing a science and risk-approach on how drug quality can be monitored and improved through integrated continuous manufacturing. This work may inform future regulatory guidelines that the pharmaceutical industry can leverage.
The Continuus team is now creating a fully automated, end-to-end, integrated continuous pilot plant that will be used to test relevant regulatory principles, including real-time release and traceability. It is expected to be completed by next year.
Continuous Processing: New Facility Requirements
Continuous processing facilities have different requirements, and therefore differing site selection needs from batch processing pharmaceutical plants. Below is a breakdown of these factors:
Facility Size/Layout: Continuous processing facilities are more vertical in design, as the process is fed from the top and product is formed as it moves through the equipment to the bottom. This means a smaller building footprint is required, with higher building heights of 60 feet or more. As finished products are free to be immediately shipped upon packaging, requirements for warehouse space are smaller than in a batch facility which requires a two-week hold on each batch for final testing.
Labor: Continuous processing facilities demand a smaller overall headcount due to decreased product handling during the process. Less lab support is needed due to real-time electronic monitoring of the product via testing instruments built into the process. The new monitoring equipment, however, triggers a requirement for IT specialists who can program software and run the analysis, and scientific statisticians who can evaluate, interpret and manage data. As with other types of advanced manufacturing, labor skill availability will be key location criteria for these plants. Transportation: As raw materials feeding the continuous process are shipped into the facility in bulk, there is an increased need for transportation access, such as rail, as well as on-site storage such as silos, according to Rooney. Inside the facility, new types of handling equipment may be required to handle the larger volume of raw materials.
Transportation: As raw materials feeding the continuous process are shipped into the facility in bulk, there is an increased need for transportation access, such as rail, as well as on-site storage such as silos, according to Rooney. Inside the facility, new types of handling equipment may be required to handle the larger volume of raw materials.
Water: Continuous processing operations require less water overall, because there are fewer individual pieces of equipment that need to be cleaned. This process also results in decreased sewer capacity requirements.
Electric Power: Moving to continuous processing has benefits related to energy usage. Pharma operations that operate in batch processing have electric power usage that fluctuates based on when processes are running a batch versus being between batches.
“Batch processing operations typically require higher electric demand that can drive rates up,” said Tim Comerford, senior vice president of energy services for BLS & Co. “Utility service providers typically prefer operations that have more consistent power demand as opposed to a fluctuating need.” This more consistent demand can also mean that requirements for electric power infrastructure at the site are also reduced, said Comerford.
A Continuous Future
So what’s next for the continuous processing method? Some drug companies are diving in full force: Janssen has reported plans to switch to continuous processing and to manufacture 70 percent of its highest volume products. Whether these will be new production plants or a retrofit of current facilities still remains to be seen. These changes will depend on two points:
1) The ability of a current facility to meet the needs of continuous operations (ceiling height could be one of the biggest issues)
2) The ability to retrain current production labor or attract new talent required to operate the equipment/ technology and to evaluate and manage the data resulting from testing instruments.
However, many pharmaceutical manufacturers are still evaluating the pros and cons. With larger companies like Roche and Merck still relying heavily on the batch release process, the future of continuous processing is one that pharmaceutical executives and facility managers should monitor closely. T&ID

